Draw The Best Lewis Structure For Ci3-1. What Is The Formal Charge On The C?
Chapter vii. Chemic Bonding and Molecular Geometry
7.iv Formal Charges and Resonance
Learning Objectives
By the terminate of this department, you will be able to:
- Compute formal charges for atoms in whatever Lewis structure
- Use formal charges to identify the most reasonable Lewis structure for a given molecule
- Explain the concept of resonance and depict Lewis structures representing resonance forms for a given molecule
In the previous section, we discussed how to write Lewis structures for molecules and polyatomic ions. As nosotros have seen, however, in some cases, at that place is seemingly more than one valid structure for a molecule. We tin can utilize the concept of formal charges to help u.s. predict the most appropriate Lewis structure when more one is reasonable.
Calculating Formal Accuse
The formal charge of an cantlet in a molecule is the hypothetical charge the cantlet would accept if we could redistribute the electrons in the bonds evenly between the atoms. Another way of saying this is that formal accuse results when nosotros take the number of valence electrons of a neutral atom, subtract the nonbonding electrons, and so subtract the number of bonds continued to that cantlet in the Lewis structure.
Thus, we calculate formal charge as follows:
[latex]\text{formal accuse} = \# \;\text{valence trounce electrons (complimentary cantlet)} \; - \;\# \;\text{lone pair electrons}\; - \frac{ane}{2} \# \;\text{bonding electrons}[/latex]
We tin double-check formal charge calculations by determining the sum of the formal charges for the whole structure. The sum of the formal charges of all atoms in a molecule must be zero; the sum of the formal charges in an ion should equal the charge of the ion.
We must remember that the formal charge calculated for an atom is not the actual charge of the cantlet in the molecule. Formal charge is only a useful bookkeeping procedure; it does non point the presence of bodily charges.
Example 1
Calculating Formal Charge from Lewis Structures
Assign formal charges to each atom in the interhalogen ion ICl4 −.
Solution
- We divide the bonding electron pairs equally for all I–Cl bonds:
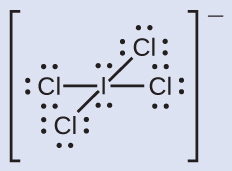
- We assign lone pairs of electrons to their atoms. Each Cl atom now has seven electrons assigned to it, and the I atom has eight.
- Subtract this number from the number of valence electrons for the neutral atom: I: seven – viii = –1Cl: seven – 7 = 0The sum of the formal charges of all the atoms equals –1, which is identical to the charge of the ion (–1).
Check Your Learning
Calculate the formal charge for each atom in the carbon monoxide molecule:

Example 2
Calculating Formal Charge from Lewis Structures
Assign formal charges to each cantlet in the interhalogen molecule BrClthree.
Solution
- Assign ane of the electrons in each Br–Cl bail to the Br atom and one to the Cl atom in that bail:
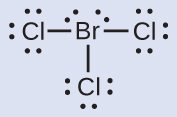
- Assign the solitary pairs to their cantlet. At present each Cl atom has vii electrons and the Br atom has 7 electrons.
- Subtract this number from the number of valence electrons for the neutral atom. This gives the formal charge:Br: 7 – 7 = 0Cl: 7 – 7 = 0
All atoms in BrCl3 have a formal accuse of naught, and the sum of the formal charges totals zero, equally it must in a neutral molecule.
Check Your Learning
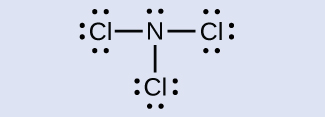
Make up one's mind the formal charge for each atom in NClthree.
Answer:
N: 0; all three Cl atoms: 0
Using Formal Charge to Predict Molecular Structure
The arrangement of atoms in a molecule or ion is called its molecular structure. In many cases, following the steps for writing Lewis structures may lead to more than one possible molecular structure—different multiple bail and lonely-pair electron placements or different arrangements of atoms, for example. A few guidelines involving formal accuse can be helpful in deciding which of the possible structures is most probable for a particular molecule or ion:
- A molecular structure in which all formal charges are zero is preferable to one in which some formal charges are non cipher.
- If the Lewis structure must have nonzero formal charges, the arrangement with the smallest nonzero formal charges is preferable.
- Lewis structures are preferable when next formal charges are goose egg or of the opposite sign.
- When nosotros must choose amidst several Lewis structures with like distributions of formal charges, the structure with the negative formal charges on the more electronegative atoms is preferable.
To encounter how these guidelines use, allow us consider some possible structures for carbon dioxide, COii. We know from our previous discussion that the less electronegative cantlet typically occupies the primal position, merely formal charges allow us to understand why this occurs. We tin can draw three possibilities for the structure: carbon in the centre and double bonds, carbon in the eye with a single and triple bond, and oxygen in the center with double bonds:
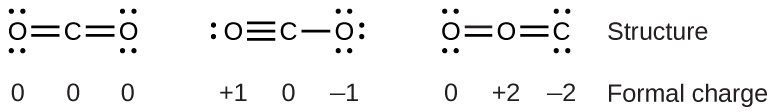
Comparison the iii formal charges, nosotros tin definitively identify the structure on the left as preferable because it has only formal charges of zero (Guideline 1).
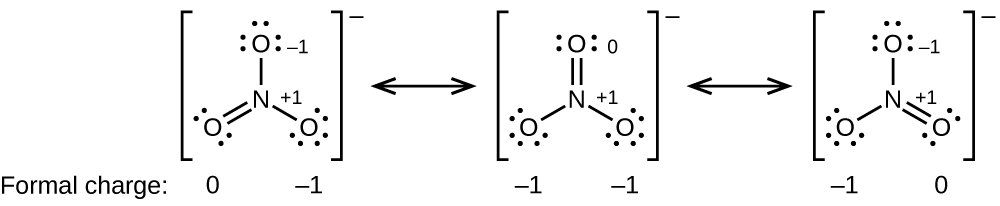
As another instance, the thiocyanate ion, an ion formed from a carbon cantlet, a nitrogen atom, and a sulfur atom, could have iii unlike molecular structures: CNS–, NCS–, or CSN–. The formal charges present in each of these molecular structures tin can aid us pick the well-nigh likely organization of atoms. Possible Lewis structures and the formal charges for each of the three possible structures for the thiocyanate ion are shown hither:
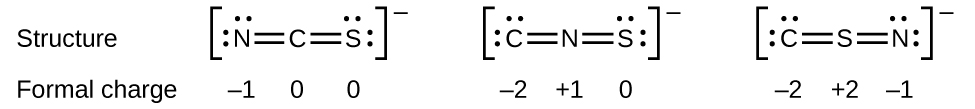
Annotation that the sum of the formal charges in each case is equal to the charge of the ion (–1). Withal, the first organisation of atoms is preferred because information technology has the lowest number of atoms with nonzero formal charges (Guideline ii). Also, it places the to the lowest degree electronegative cantlet in the centre, and the negative charge on the more electronegative chemical element (Guideline 4).
Instance 3
Using Formal Accuse to Determine Molecular Structure
Nitrous oxide, N2O, commonly known every bit laughing gas, is used as an coldhearted in minor surgeries, such as the routine extraction of wisdom teeth. Which is the likely construction for nitrous oxide?
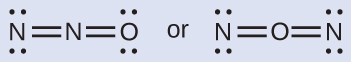
Solution
Determining formal charge yields the following:

The structure with a concluding oxygen atom best satisfies the criteria for the most stable distribution of formal accuse:
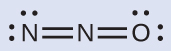
The number of atoms with formal charges are minimized (Guideline 2), and in that location is no formal charge larger than i (Guideline 2). This is once more consequent with the preference for having the less electronegative cantlet in the central position.
Check Your Learning
Which is the nigh probable molecular structure for the nitrite (NOii −) ion?
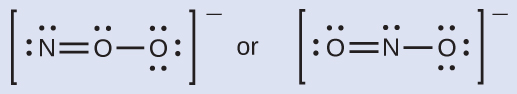
Resonance
You may accept noticed that the nitrite anion in Example 3 can have ii possible structures with the atoms in the same positions. The electrons involved in the N–O double bail, however, are in different positions:
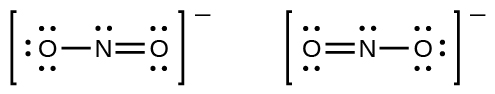
If nitrite ions exercise indeed comprise a single and a double bail, we would wait for the two bond lengths to be different. A double bond between two atoms is shorter (and stronger) than a single bail between the same two atoms. Experiments prove, still, that both Due north–O bonds in NO2 − have the same strength and length, and are identical in all other properties.
Information technology is non possible to write a unmarried Lewis construction for NO2 − in which nitrogen has an octet and both bonds are equivalent. Instead, nosotros use the concept of resonance: if ii or more than Lewis structures with the same arrangement of atoms can be written for a molecule or ion, the bodily distribution of electrons is an average of that shown by the various Lewis structures. The actual distribution of electrons in each of the nitrogen-oxygen bonds in NOtwo − is the boilerplate of a double bond and a unmarried bail. We call the individual Lewis structures resonance forms. The actual electronic structure of the molecule (the average of the resonance forms) is called a resonance hybrid of the individual resonance forms. A double-headed pointer between Lewis structures indicates that they are resonance forms. Thus, the electronic construction of the NO2 − ion is shown equally:
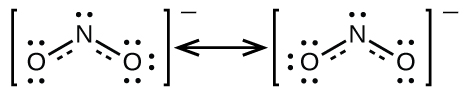
We should remember that a molecule described as a resonance hybrid never possesses an electronic structure described by either resonance form. It does not fluctuate betwixt resonance forms; rather, the actual electronic structure is ever the boilerplate of that shown by all resonance forms. George Wheland, one of the pioneers of resonance theory, used a historical analogy to draw the human relationship between resonance forms and resonance hybrids. A medieval traveler, having never before seen a rhinoceros, described it as a hybrid of a dragon and a unicorn because it had many backdrop in common with both. Only as a rhinoceros is neither a dragon sometimes nor a unicorn at other times, a resonance hybrid is neither of its resonance forms at whatever given time. Similar a rhinoceros, it is a real entity that experimental evidence has shown to be. It has some characteristics in common with its resonance forms, only the resonance forms themselves are convenient, imaginary images (like the unicorn and the dragon).
The carbonate anion, CO3 2−, provides a second example of resonance:
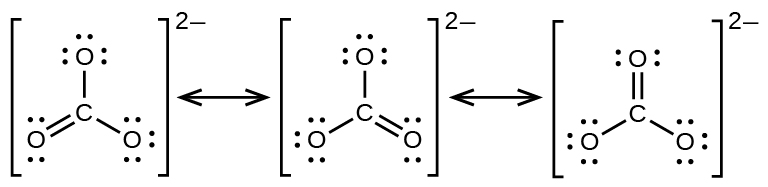
One oxygen atom must accept a double bail to carbon to complete the octet on the central atom. All oxygen atoms, however, are equivalent, and the double bond could form from any one of the three atoms. This gives rise to three resonance forms of the carbonate ion. Because we tin write three identical resonance structures, nosotros know that the bodily organization of electrons in the carbonate ion is the boilerplate of the three structures. Again, experiments show that all iii C–O bonds are exactly the same.

The online Lewis Structure Make includes many examples to practice drawing resonance structures.
Key Concepts and Summary
In a Lewis structure, formal charges can be assigned to each cantlet past treating each bond as if i-half of the electrons are assigned to each atom. These hypothetical formal charges are a guide to determining the well-nigh appropriate Lewis structure. A construction in which the formal charges are every bit close to nix as possible is preferred. Resonance occurs in cases where ii or more than Lewis structures with identical arrangements of atoms but different distributions of electrons can be written. The actual distribution of electrons (the resonance hybrid) is an average of the distribution indicated by the individual Lewis structures (the resonance forms).
Key Equations
- [latex]\text{formal accuse} = \# \;\text{valence shell electrons (free atom)} \; - \;\# \;\text{lone pair electrons}\; - \frac{1}{2} \# \;\text{bonding electrons}[/latex]
Chemistry End of Chapter Exercises
- Write resonance forms that describe the distribution of electrons in each of these molecules or ions.
(a) selenium dioxide, OSeO
(b) nitrate ion, NO3 −
(c) nitric acid, HNO3 (N is bonded to an OH grouping and two O atoms)
(d) benzene, CsixH6:
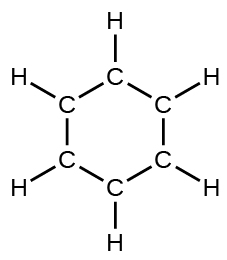
(eastward) the formate ion:
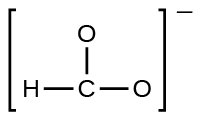
- Write resonance forms that depict the distribution of electrons in each of these molecules or ions.
(a) sulfur dioxide, So2
(b) carbonate ion, COthree ii−
(c) hydrogen carbonate ion, HCO3 − (C is bonded to an OH group and two O atoms)
(d) pyridine:
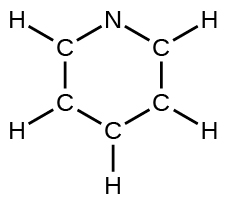
(e) the allyl ion:
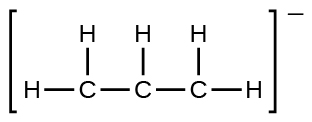
- Write the resonance forms of ozone, O3, the component of the upper atmosphere that protects the Earth from ultraviolet radiation.
- Sodium nitrite, which has been used to preserve bacon and other meats, is an ionic chemical compound. Write the resonance forms of the nitrite ion, NO2 –.
- In terms of the bonds present, explain why acetic acid, CH3CO2H, contains two distinct types of carbon-oxygen bonds, whereas the acetate ion, formed past loss of a hydrogen ion from acetic acrid, only contains one type of carbon-oxygen bond. The skeleton structures of these species are shown:
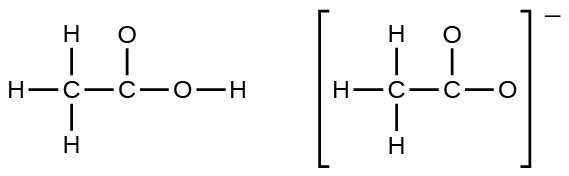
- Write the Lewis structures for the following, and include resonance structures where appropriate. Indicate which has the strongest carbon-oxygen bond.
(a) CO2
(b) CO
- Toothpastes containing sodium hydrogen carbonate (sodium bicarbonate) and hydrogen peroxide are widely used. Write Lewis structures for the hydrogen carbonate ion and hydrogen peroxide molecule, with resonance forms where appropriate.
- Make up one's mind the formal accuse of each element in the following:
(a) HCl
(b) CF4
(c) PCl3
(d) PF5
- Determine the formal charge of each element in the following:
(a) H3O+
(b) SO4 2−
(c) NH3
(d) Otwo two−
(e) H2Otwo
- Calculate the formal charge of chlorine in the molecules Cltwo, BeCl2, and ClFv.
- Calculate the formal charge of each element in the following compounds and ions:
(a) F2CO
(b) NO–
(c) BFfour −
(d) SnClthree −
(eastward) H2CCHtwo
(f) ClF3
(g) SeFvi
(h) PO4 3−
- Draw all possible resonance structures for each of these compounds. Determine the formal charge on each cantlet in each of the resonance structures:
(a) O3
(b) SOii
(c) NO2 −
(d) NO3 −
- Based on formal charge considerations, which of the following would probable exist the correct arrangement of atoms in nitrosyl chloride: ClNO or ClON?
- Based on formal charge considerations, which of the following would likely exist the correct arrangement of atoms in hypochlorous acid: HOCl or OClH?
- Based on formal charge considerations, which of the post-obit would likely be the correct arrangement of atoms in sulfur dioxide: OSO or SOO?
- Draw the structure of hydroxylamine, HthreeNO, and assign formal charges; look up the construction. Is the actual construction consistent with the formal charges?
- Iodine forms a series of fluorides (listed hither). Write Lewis structures for each of the four compounds and determine the formal charge of the iodine atom in each molecule:
(a) IF
(b) IF3
(c) IFfive
(d) IFvii
- Write the Lewis construction and chemical formula of the compound with a molar mass of about 70 yard/mol that contains 19.7% nitrogen and 80.iii% fluorine past mass, and make up one's mind the formal charge of the atoms in this chemical compound.
- Which of the following structures would we expect for nitrous acid? Determine the formal charges:
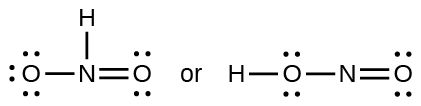
- Sulfuric acid is the industrial chemical produced in greatest quantity worldwide. About 90 billion pounds are produced each year in the The states solitary. Write the Lewis structure for sulfuric acrid, H2SOiv, which has two oxygen atoms and two OH groups bonded to the sulfur.
Glossary
- formal accuse
- charge that would outcome on an atom by taking the number of valence electrons on the neutral atom and subtracting the nonbonding electrons and the number of bonds (half of the bonding electrons)
- molecular construction
- arrangement of atoms in a molecule or ion
- resonance
- situation in which one Lewis structure is insufficient to draw the bonding in a molecule and the average of multiple structures is observed
- resonance forms
- two or more Lewis structures that have the same arrangement of atoms but different arrangements of electrons
- resonance hybrid
- boilerplate of the resonance forms shown by the individual Lewis structures
Solutions
Answers to Chemistry Terminate of Affiliate Exercises
2. (a)
(b)
(c)
(d)
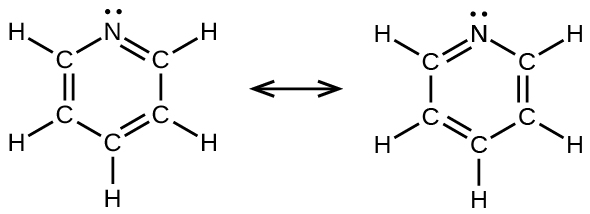
(e)
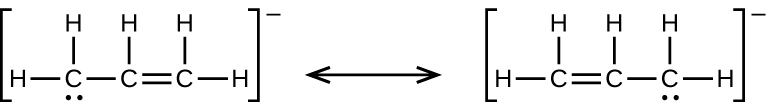
4.
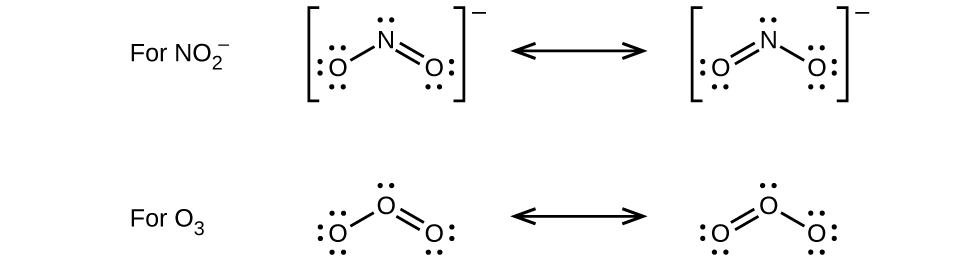
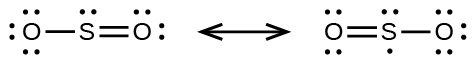
6. (a)
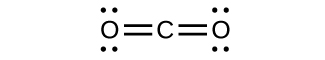
(b)
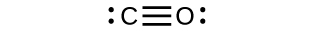
CO has the strongest carbon-oxygen bond because there is a triple bail joining C and O. CO2 has double bonds.
eight. (a) H: 0, Cl: 0; (b) C: 0, F: 0; (c) P: 0, Cl 0; (d) P: 0, F: 0
ten. Cl in Cltwo: 0; Cl in BeCl2: 0; Cl in ClF5: 0
12. (a)
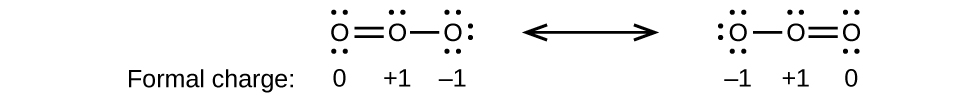 ;
;
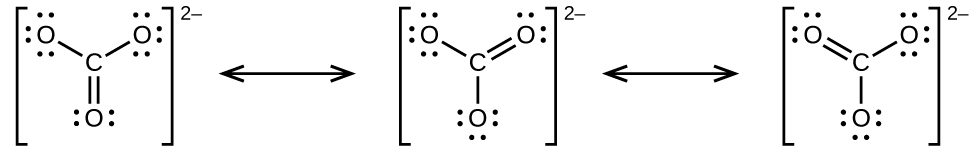
(b)
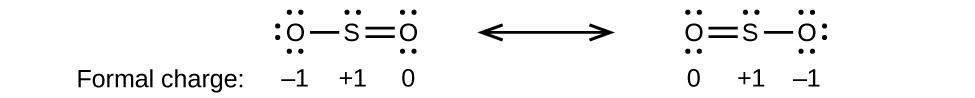 ;
;
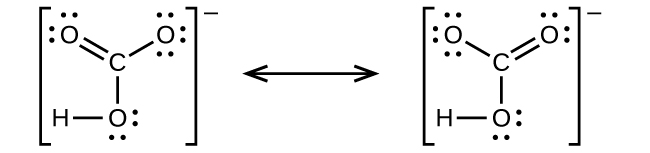
(c)
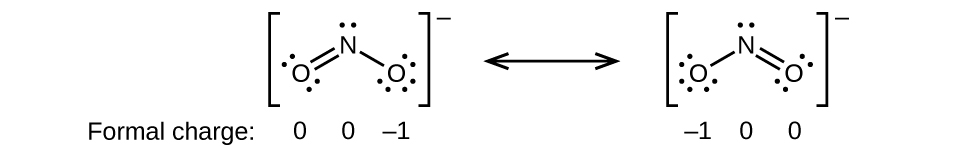 ;
;
(d)
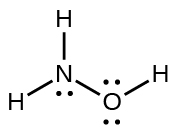
14. HOCl
16. The structure that gives zero formal charges is consistent with the actual structure:
eighteen. NFiii;
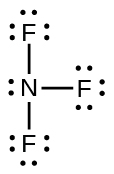
20.
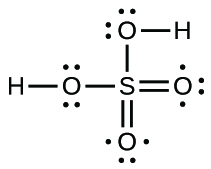
Source: https://opentextbc.ca/chemistry/chapter/7-4-formal-charges-and-resonance/
Posted by: rickerwhimere.blogspot.com


0 Response to "Draw The Best Lewis Structure For Ci3-1. What Is The Formal Charge On The C?"
Post a Comment